Hey there! As a supplier of aliphatic heterocycles, I've seen firsthand the importance of maximizing the bioavailability of these drugs. Bioavailability is a big deal because it determines how much of a drug actually gets into the bloodstream and reaches its target site in the body. If a drug has low bioavailability, it might not work as well as it should, or you might need to take higher doses, which can lead to more side effects. So, let's dig into how we can boost the bioavailability of aliphatic heterocyclic drugs.
Understanding Aliphatic Heterocyclic Drugs
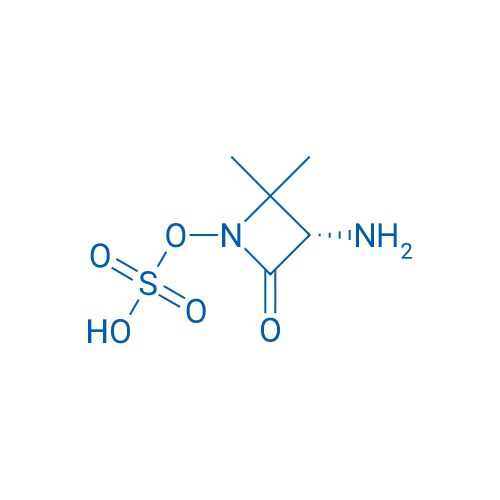
First off, what are aliphatic heterocyclic drugs? They're a group of compounds that have a cyclic structure with at least one non - carbon atom (like nitrogen, oxygen, or sulfur) in the ring, and the ring isn't part of an aromatic system. These drugs are used in a wide range of medical treatments, from antibiotics to antidepressants. For example, the (S)-3-Amino-2,2-dimethyl-4-oxoazetidin-1-yl Hydrogen Sulfate (S)-3-Amino-2,2-dimethyl-4-oxoazetidin-1-yl Hydrogen Sulfate is an important aliphatic heterocyclic compound that plays a role in certain therapeutic applications.
Factors Affecting Bioavailability
Before we talk about solutions, we need to understand what factors can mess with the bioavailability of these drugs.
Absorption in the Gut
Most drugs are absorbed in the gastrointestinal (GI) tract. The solubility of the drug in the GI fluids is crucial. If a drug is poorly soluble, it won't dissolve well in the gut, and less of it will be absorbed into the bloodstream. Also, the pH of the GI tract can affect the ionization state of the drug. Aliphatic heterocyclic drugs can be acidic, basic, or neutral, and their ionization state affects how easily they can cross the gut wall.
Metabolism
The liver is like a big chemical factory in our body. When a drug enters the liver, enzymes can break it down into different compounds. This is called metabolism. Some aliphatic heterocyclic drugs are rapidly metabolized in the liver, which means that a large portion of the drug is changed before it can reach its target site. This is known as the first - pass effect.
Drug Formulation
The way a drug is formulated can also have a huge impact on its bioavailability. For example, if a drug is in a tablet form, how quickly the tablet disintegrates and releases the drug can affect absorption.
Strategies to Increase Bioavailability
Solubility Enhancement
One of the most effective ways to improve bioavailability is to increase the solubility of the drug.
Salt Formation
Making a salt of the aliphatic heterocyclic drug can often increase its solubility. When a drug is in its salt form, it can dissolve more easily in water. For example, some basic aliphatic heterocyclic drugs can form salts with acids, and these salts are more soluble in the slightly acidic environment of the stomach. Take 3-(Methylsulfonyl)azetidine Hydrochloride, 3-(Methylsulphonyl)azetidine HCl (3-(Methylsulfonyl)azetidine Hydrochloride, 3-(Methylsulphonyl)azetidine HCl. The hydrochloride salt form can enhance its solubility and thus improve its bioavailability.

Use of Solubilizing Agents
We can also use solubilizing agents like surfactants. Surfactants can form micelles in solution, and the drug can be trapped inside these micelles. This increases the effective solubility of the drug. For example, polysorbate 80 is a commonly used surfactant that can improve the solubility of many poorly soluble drugs.
Prodrug Approach
A prodrug is a modified form of the original drug. It's designed to be inactive when administered but is converted into the active drug in the body. This approach can help overcome some of the problems related to absorption and metabolism. For example, by modifying the structure of an aliphatic heterocyclic drug to make a prodrug, we can improve its solubility in the GI tract or reduce its susceptibility to first - pass metabolism. The (6R,7R)-Benzhydryl 7 - amino - 3-(chloromethyl)-8 - oxo - 5 - thia - 1 - azabicyclo[4.2.0]oct - 2 - ene - 2 - carboxylate Hydrochloride (6R,7R)-Benzhydryl 7-amino-3-(chloromethyl)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate Hydrochloride could potentially be developed into a prodrug to enhance its bioavailability.
Nanoparticle Formulation
Nanoparticles are tiny particles, usually in the range of 1 - 1000 nanometers. When a drug is formulated into nanoparticles, it can have several advantages. Nanoparticles can increase the surface area of the drug, which improves its solubility and dissolution rate. They can also protect the drug from degradation in the GI tract and the liver. For example, lipid - based nanoparticles can encapsulate the drug and improve its delivery to the target site.
Modified Release Formulations
Modified release formulations are designed to release the drug slowly over time. This can be beneficial for aliphatic heterocyclic drugs because it can maintain a steady concentration of the drug in the bloodstream. There are two main types: sustained release and controlled release. Sustained release formulations release the drug at a relatively constant rate over a long period, while controlled release formulations can be designed to release the drug at specific times or in response to certain stimuli.
The Role of Our Company
As a supplier of aliphatic heterocycles, we play a crucial role in helping our customers develop drugs with better bioavailability. We can provide high - quality raw materials for drug development. Our team of experts can also offer technical support on solubility enhancement, prodrug design, and formulation development. We understand that every drug is unique, and we're committed to working closely with our customers to find the best solutions for their specific needs.
If you're in the pharmaceutical industry and are looking to develop aliphatic heterocyclic drugs with improved bioavailability, we'd love to talk to you. Whether you need advice on formulation, high - quality raw materials, or just want to discuss the latest trends in drug development, we're here to help. Reach out to us to start a conversation about your project. We're confident that our products and expertise can make a difference in your drug development process.
References
- Stella, V. J., & Nti - Addae, K. (2007). Prodrugs: A chemical approach to improve drug delivery. In Encyclopedia of pharmaceutical technology.
- Lobenberg, R., & Amidon, G. L. (2000). Modern bioavailability, bioequivalence and biopharmaceutics classification system. New scientific approaches to international regulatory standards. Eur J Pharm Sci, 11 Suppl 2, S61 - 80.
- Allen, L. V., & Popovich, N. G. (2013). Ansel's pharmaceutical dosage forms and drug delivery systems. Lippincott Williams & Wilkins.