| Product Name | 2,2,3,3,3-Pentafluoropropyl trifluoromethanesulfonate |
| CAS Number | 6401-00-9 |
| Molecular Formula | C4H2F8O3S |
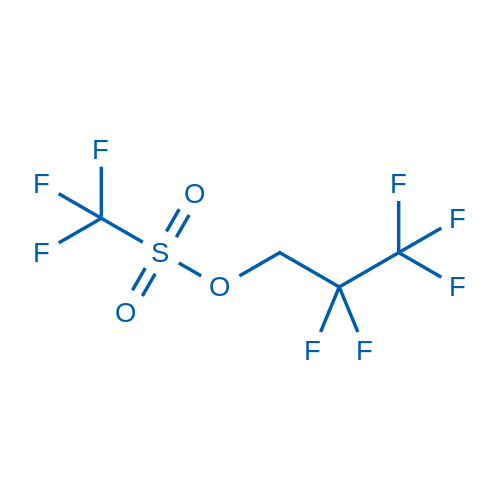
| Linear Formula | CF3SO2OCH2CF2CF3 |
| Molecular Weight | 282.11 |
| SMILES Code | O=S(C(F)(F)F)(OCC(F)(F)C(F)(F)F)=O |
| MDL No. | MFCD01862006 |
| Pubchem ID | 2776019 |
| InChI Key | QHZUUSNAUOEJBB-UHFFFAOYSA-N |
Chemical Properties
This compound is a dense(1.702 g/mL),volatile liquid with a boiling point of 102-105°C.It is a highly fluorinated alkylating agent characterized by extreme moisture sensitivity-the triflate group hydrolyzes rapidly in air.It is miscible with many organic solvents but reacts violently with water,alcohols,and amines.It must be stored under strict anhydrous conditions,often over molecular sieves,and handled in a well-ventilated fume hood due to potential HF release upon decomposition.
Description
2,2,3,3,3-Pentafluoropropyl trifluoromethanesulfonate is a superior perfluoroalkylating reagent.It combines a highly electron-withdrawing,lipophilic pentafluoropropyl group with an exceptionally good leaving group(triflate).This makes it an immensely powerful electrophile for introducing the-CH₂CF₂CF₃moiety,a motif known to dramatically enhance metabolic stability,lipophilicity,and membrane permeability in bioactive molecules-a property often termed the"pentafluoropropyl effect."
Uses
1.Pharmaceutical Synthesis
Crucial for the late-stage pentafluoropropylation of drug candidates,particularly in medicinal chemistry programs aimed at improving metabolic stability and brain penetration.It is used to alkylate O-,N-,S-,and C-nucleophiles in targets for CNS diseases and oncology.
2.Agrochemical R&D
Employed to incorporate the pentafluoropropyl group into novel pesticides,creating analogs with improved rainfastness,vapor pressure,and resistance to metabolic degradation by target pests,leading to longer-lasting field effects.
3.Functional Material Synthesis
Used in surface science to create superhydrophobic and oleophobic coatings.It can react with surface hydroxyl groups on glass,metals,or polymers to graft the low-surface-energy pentafluoropropyl chain,imparting stain-resistant and easy-clean properties.
4.Organic Synthesis Building Block
A benchmark reagent for studying nucleophilic substitution kinetics with super-leaving groups.It is also a key tool in the synthesis of fluorinated dendrimers,polymers,and liquid crystals,where the-CH₂CF₂CF₃group influences phase behavior and optical properties.





![2-(1-(2-Fluorobenzyl)piperidin-4-yl)-1H-benzo[d]imidazole](/uploads/44503/small/2-1-2-fluorobenzyl-piperidin-4-yl-1h-benzo-d21290.png?size=195x0)




![Methyl 5-(hydroxymethyl)bicyclo[3.1.1]heptane-1-carboxylate](/uploads/44503/small/methyl-5-hydroxymethyl-bicyclo-3-1-1-heptanee2736.png?size=195x0)